The calculator will autopopulate other fields. NOTE: Fill in at least two values to obtain the result of another by clicking the calculate button. Well 1 3.64E-03 1.81E-03 2.74E-03 5. Molality similar to molarity, however calculation of molality uses the mass rather than volume of the solvent used, making it temperature independent unlike molarity. (ca2+) Ion activity (SO+4) Ion activity (ca2+) Ion activity (SO+4) Ion activity Percent error, 100*(Cat-|An|)/(Cat+|An|) = -19.91įor gypsum activity for Ca+2 and SO4 are important to me, Ca activity is not that different, but SO4 really is as can you see from the table below. Formula: Molarity ( Parts Per Million (PPM) value × 0. Conversion to molarity units requires only the molecular weight. I'm trying to get the saturation index for gypsum for 60 wells, I uploaded my Data into the solution spreadsheet, once in ppm (mg/L) and again in Molarity (mol/L) and run both programs, theoretically the saturation index should be the same for both runs, but its not, mainly the activity for SO4 is different among other things, for example, the "solution description" below shows how they give different results: PPM to Molarity Formula: To find the molarity from PPM, first multiply the PPM value with 0.001 and divide the resultant value by atomic weight. To convert to ppm, which is a mass/mass unit, it is necessary to convert the volume of. Of moles of that component present in the solution to the total number of molesĬonsider a binary solution components solvent (A) and solute (B).Thank you for this helpful forum, I'm using PHREEQC for my groundwater research, and I came upon this issue:

The Relation Between Mole Fraction and Molality:įraction of any component of a solution is defined as the ratio of the number The formality of a solution may be defined as a number of moles of ionic solute Ionic compound is called formole and its molarity is called formality. This concept is used in the case of ionic substances. The formality of a solution depends on temperature. Once you have mg/L, it is much easier to convert out to other more useful concentrations like molarity (mol/L) which is needed for all aqueous equilibrium.
#How to calculate ppm with molarity how to#
If the formula mass of solute is equal to its molar mass, then the formality is equal to molarity. This chemistry video tutorial explains how to convert the solution concentration from parts per million or ppm to Molarity.My Website.
The number of formula mass in gram present per litre of a solution.
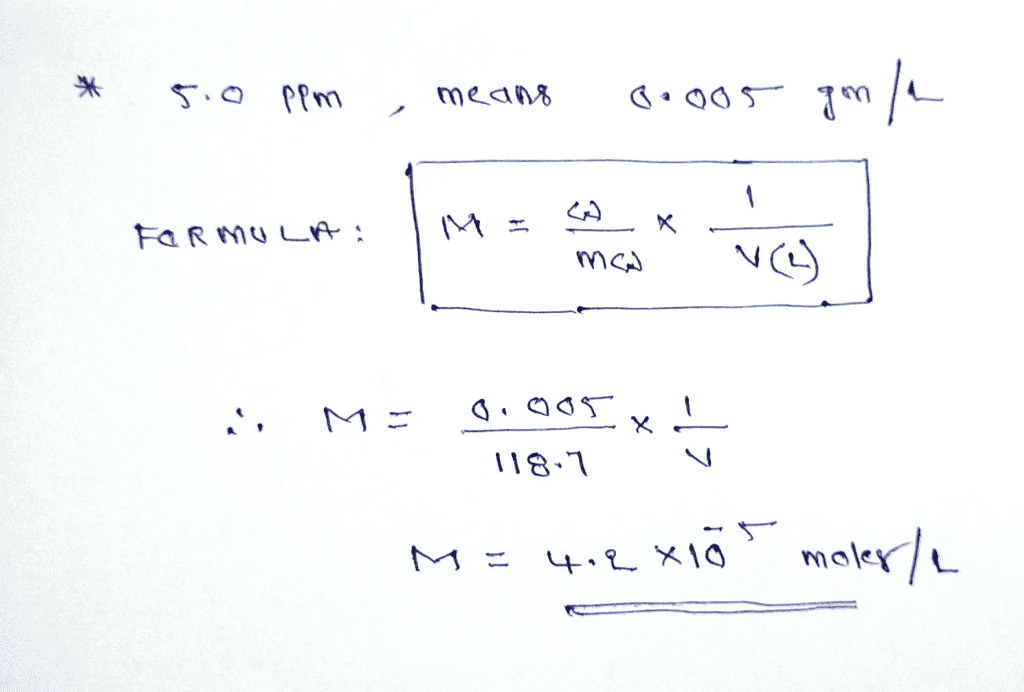
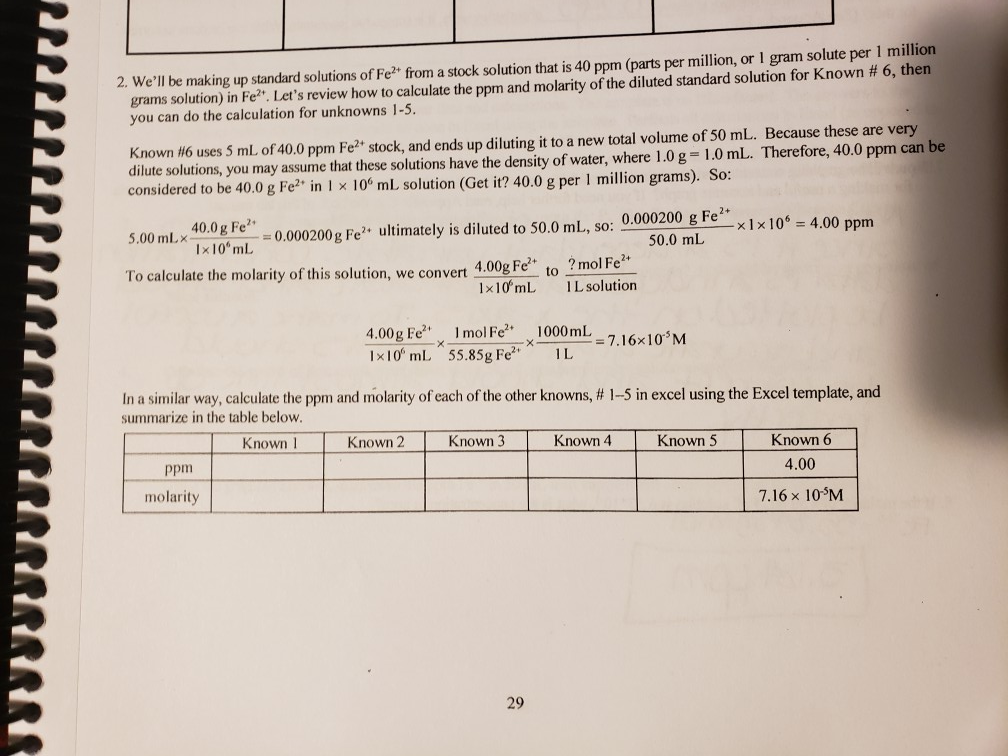
Our online molarity to PPM calculator will help you to convert more than 100 different types of elements from molarity to ppm considering the atomic mass of the typical element. stock solution that is 40 ppm (parts per million, or I gram solute per 1 million grams solutionin Fe Lets review howto calculate the ppmi and molarity. The equation for Beer's law is: A mCl (Aabsorbance, m molar extinction coefficient. You will be applying Beer's law to calculate the concentration. Once you have that you can compare the absorbance value of an unknown sample to figure out its concentration. A solution having normality equal to unity is called a normal solution.Įquivalent mass = strength of solution in g/L. Molarity to PPM Calculator Parts per million also known as PPM is a used for measuring the substance quantity per million parts of solution. A mCl The basic idea here is to use a graph plotting Absorbance vs.
